 One of them simply involves the fact that there are two interactions going on here. The BornLand equation (Equation 21.5.5) is a means of calculating the lattice energy of a crystalline ionic compound and derived from the electrostatic potential of the ionic lattice and a repulsive potential energy term. Lattice energy depends on the strength of interactions between cations and anions in the lattice, which we can estimate using Coulomb's law: F (qq)/r. 
In this case, there are a couple of other factors, some of which are more complicated. The energy required to separate the ions in a crystal lattice into individual gaseous ions is known as lattice energy. This sort of behavior, in which we start to see a trend but it then reverses, often means there is more than one factor at work. The idea is to show what factors are involved when an alkaline earth metal forms stable compounds with MX2+ M X 2 + ions whereas an alkali metal favors MX+ M X +. Once again, it isn't surprising that the lithium chloride is the least soluble, but the most soluble seems to be the lithium bromide, not the lithium iodide. (iii) The lattice energy affects the solubility of the ionic compounds.\) Water solubility among lithium halides. (ii) The lattice energy is greater for small, highly charged ions. They will have the smallest distance between centres and will have the largest lattice energies. (i) The greater the lattice enthalpy, the more the stability of ionic compounds. NaF crystallizes in the same structure as LiF but with a NaF distance of 231 pm. The important consequence of the lattice energies are: The lattice energy of LiF is 1023 kJ/mol, and the LiF distance is 200.8 pm. $\therefore $The order of lattice energy is $NaF > NaCl > KCl > RbCl$. Electron gain enthalpy of chlorine is most negative so it has more tendency to gain. The given ionic solids are $NaCl,$ion so $NaCl$has greater lattice energy than $KCl$ and $RbCl$. NaCl is more ionic because chlorine is more electronegative than iodine. Thus, the lattice energy decreases in the order 'LiCl > KF > NaCl > NaBr' 747color(white)(mll)786color(white)(mm)821color(white)(mml)853color(white)(l) 'kJ·mol''-1' Just to confirm our predictions, I have listed the actual lattice energies below the formulas. F- is smaller than Cl- so lattice energy is higher. Hence, more will be the value of lattice energy. In other words, the greater the interionic distance, the smaller the lattice energy. Lattice energy is higher in NaF because what matters is distance to closest ion which is Na-F distance. 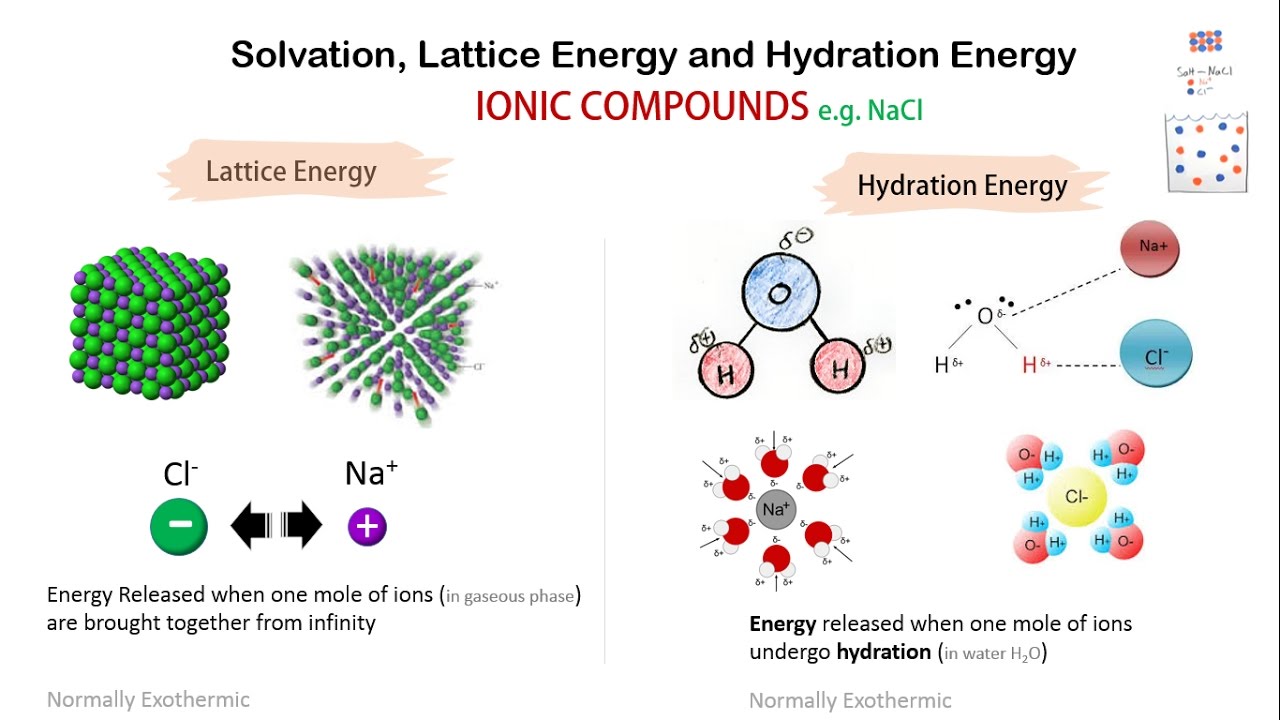
From Hess' Law: Hf s + 12D + IP + EA +EL 2RT 396 kJmol H f s + 1 2 D + I P + E A + E L 2 R T 396 k J m o l. smaller the size of ions, lesser is the internuclear distance and greater will be the interionic attraction. We can alternatively construct a Born-Haber cycle for the formation of NaCl from the elements and calculate the lattice energy as the 'missing' term in the cycle. Hence the correct order of increasing lattice. Charge density of MgO is greater compared to NaF, so MgO has a higher lattice energy. Oxygen carries a 2- charge while Cl carries a 1- charge. The lattice energies of NaCl, NaF, KCl and RbCl follow the order 1. Mg carries a 2+ charge while Na carries a 1+ charge. The lattice energy of MgO is greater than NaF. larger the magnitude of charge on ions, greater will be the attractive forces, higher is the value of lattice energy and inversely proportional to size of an ion i.e. The charge density is high in AlN than in MgO and NaF. Lattice energy is directly proportional to charge on ion i.e. 
Lattice energy depends upon charge and size of cation. But as Al3+ has the smallest size wouldn't three F- electrons result in very large magnitude of inter electronic repulsion, thus decreasing its. The lattice energy of NaCl, for example, is 787. As atomic size decreases across a period, the order of stability is AlF3 > MgF2> NaF. The lattice energies of ionic compounds are relatively large. RbCl has NaCl type lattice and its unit cell length is 0.30A greater than that for KCl. Na + ( g) + Cl - ( g) NaCl ( s) Ho -787.3 kJ/mol. The correct order of increasing lattice energy for: Answer whether the above statement is true or false. 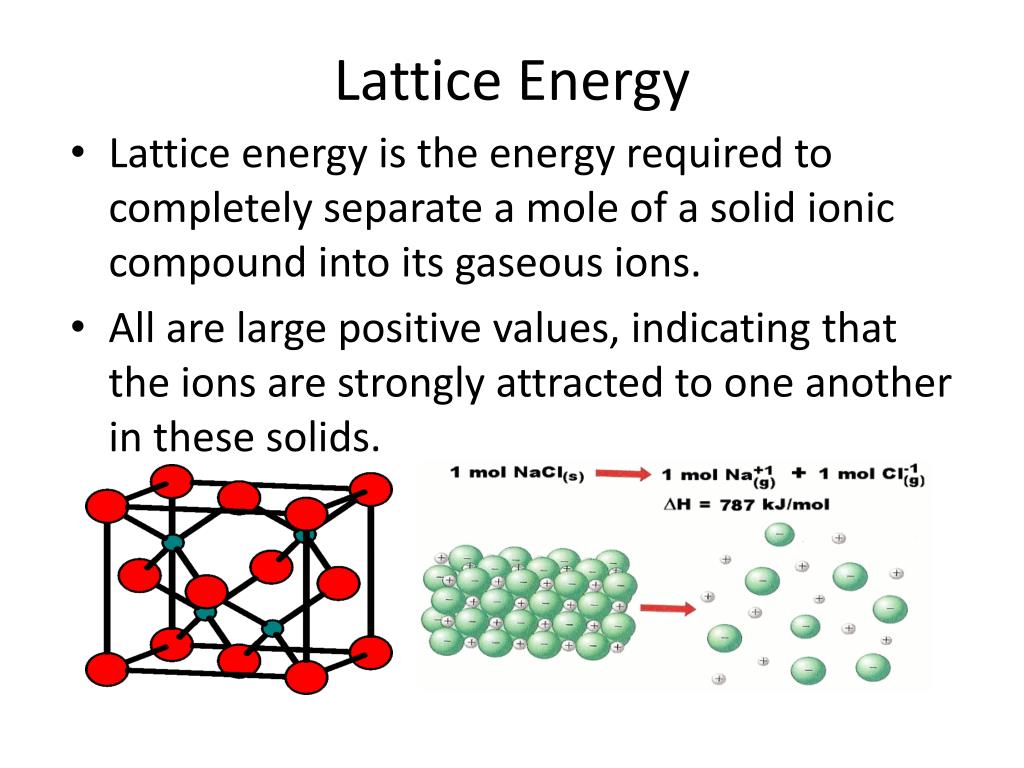
Hint: Lattice energy: The amount of energy released when $1$mole of an ionic solid is formed from its gaseous ions. The lattice energies of N aCl, N aF, KCl and RbCl follow the order: Q. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
